“人工细胞”的版本间的差异
| 第22行: | 第22行: | ||
自下而上构建人工细胞的方法涉及创建一个完全由非生物材料构成的原生质体。正如术语“细胞”所暗示的那样,一个前提是要生成某种定义了单个细胞单元的隔室。由于磷脂膜在所有活的生物细胞中起着选择性屏障的作用,<ref>{{cite journal | vauthors = Kamiya K, Takeuchi S | title = Giant liposome formation toward the synthesis of well-defined artificial cells | journal = Journal of Materials Chemistry B | volume = 5 | issue = 30 | pages = 5911–5923 | date = August 2017 | pmid = 32264347 | doi = 10.1039/C7TB01322A }}</ref> 因此可以选择磷脂膜作为一个明显的划分界限。科学家通过将生物分子包裹在细胞大小的磷脂小泡中,观察这些分子在生物细胞中的类似作用,从而重建某些细胞功能。<ref>{{cite journal | vauthors = Litschel T, Schwille P | title = Protein Reconstitution Inside Giant Unilamellar Vesicles | journal = Annual Review of Biophysics | volume = 50 | pages = 525–548 | date = May 2021 | pmid = 33667121 | doi = 10.1146/annurev-biophys-100620-114132 | s2cid = 232131463 }}</ref>类似地,功能性生物构建模块可以被封装在这些脂质隔室中,以实现人工细胞的合成(无论多么初级)。 | 自下而上构建人工细胞的方法涉及创建一个完全由非生物材料构成的原生质体。正如术语“细胞”所暗示的那样,一个前提是要生成某种定义了单个细胞单元的隔室。由于磷脂膜在所有活的生物细胞中起着选择性屏障的作用,<ref>{{cite journal | vauthors = Kamiya K, Takeuchi S | title = Giant liposome formation toward the synthesis of well-defined artificial cells | journal = Journal of Materials Chemistry B | volume = 5 | issue = 30 | pages = 5911–5923 | date = August 2017 | pmid = 32264347 | doi = 10.1039/C7TB01322A }}</ref> 因此可以选择磷脂膜作为一个明显的划分界限。科学家通过将生物分子包裹在细胞大小的磷脂小泡中,观察这些分子在生物细胞中的类似作用,从而重建某些细胞功能。<ref>{{cite journal | vauthors = Litschel T, Schwille P | title = Protein Reconstitution Inside Giant Unilamellar Vesicles | journal = Annual Review of Biophysics | volume = 50 | pages = 525–548 | date = May 2021 | pmid = 33667121 | doi = 10.1146/annurev-biophys-100620-114132 | s2cid = 232131463 }}</ref>类似地,功能性生物构建模块可以被封装在这些脂质隔室中,以实现人工细胞的合成(无论多么初级)。 | ||
| − | |||
| + | 人们想到利用合成遗传信息,制备具有DNA自复制能力的磷脂双层囊泡。脂质膜的形成、通过模板过程进行的 DNA 和 RNA 复制、为了在脂膜上进行主动运输的化学能量的收集,是这种人工细胞的三个基本元素。<ref>{{cite journal | vauthors = Szostak JW, Bartel DP, Luisi PL | title = Synthesizing life | journal = Nature | volume = 409 | issue = 6818 | pages = 387–390 | date = January 2001 | pmid = 11201752 | doi = 10.1038/35053176 | s2cid = 4429162 }}</ref><ref>{{cite journal | vauthors = Pohorille A, Deamer D | title = Artificial cells: prospects for biotechnology | journal = Trends in Biotechnology | volume = 20 | issue = 3 | pages = 123–128 | date = March 2002 | pmid = 11841864 | doi = 10.1016/S0167-7799(02)01909-1 | hdl = 2060/20020043286 }}</ref>这个原始细胞将会遇到的主要障碍是创建一个最小的包含所有足够的生命信息的合成DNA,以及细胞发展中不可或缺的非遗传成分的复制,如分子自我组织。<ref>{{cite journal | vauthors = Noireaux V, Maeda YT, Libchaber A | title = Development of an artificial cell, from self-organization to computation and self-reproduction | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 108 | issue = 9 | pages = 3473–3480 | date = March 2011 | pmid = 21317359 | pmc = 3048108 | doi = 10.1073/pnas.1017075108 | doi-access = free | bibcode = 2011PNAS..108.3473N }}</ref> 不过,人们希望通过这种自下而上的方法,更深入了解关于在细胞层面组织的基本问题和生物生命起源问题。迄今为止,还没有利用生命分子合成出能够自我繁殖的完全人工细胞,尽管目前各种团体正朝着这一目标努力,但它仍然在遥远的未来。<ref>{{cite journal | vauthors = Rasmussen S, Chen L, Nilsson M, Abe S | title = Bridging nonliving and living matter | journal = Artificial Life | volume = 9 | issue = 3 | pages = 269–316 | date = Summer 2003 | pmid = 14556688 | doi = 10.1162/106454603322392479 | s2cid = 6076707 | citeseerx = 10.1.1.101.1606 }}</ref> | ||
| − | |||
| − | + | 另一种方法是,创造一个类似于人们认为在进化过程中存在的,称为原始汤的状态的原始细胞。各种RNA聚合物可以被包封在囊泡中,在如此小的边界条件下,将进行化学反应测试。<ref>{{cite journal| vauthors = Gilbert W |title=Origin of life: The RNA world|journal=Nature|date=20 February 1986 |volume=319 |issue=6055 |pages=618 |doi=10.1038/319618a0 |bibcode=1986Natur.319..618G |s2cid=8026658 }}</ref> | |
| − | |||
| − | === | + | ===伦理和争议=== |
| − | + | 原始细胞的研究引起了争议和反对意见,包括对”人工生命”模糊定义的批评。<ref>{{cite journal | vauthors = Bedau M, Church G, Rasmussen S, Caplan A, Benner S, Fussenegger M, Collins J, Deamer D | display-authors = 6 | title = Life after the synthetic cell | journal = Nature | volume = 465 | issue = 7297 | pages = 422–424 | date = May 2010 | pmid = 20495545 | doi = 10.1038/465422a | s2cid = 27471255 | bibcode = 2010Natur.465..422. }}</ref>尽管对原细胞最普遍的担忧是它们通过不受控制的复制对人类健康和环境的潜在威胁,但创造一个基本的生命单位最紧迫的问题是伦理。<ref name='Beadau'/> | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| + | ===国际研究=== | ||
2010年代中期,研究界开始认识到有必要统一人工合成细胞的研究领域,承认从非生物成分构建一个完整生物体的任务超出了一个国家的资源范围。<ref>{{cite web|url=https://www.statnews.com/2017/07/28/cell-build-from-scratch/|title=From chemicals to life: Scientists try to build cells from scratch|access-date=4 Dec 2019}}</ref> | 2010年代中期,研究界开始认识到有必要统一人工合成细胞的研究领域,承认从非生物成分构建一个完整生物体的任务超出了一个国家的资源范围。<ref>{{cite web|url=https://www.statnews.com/2017/07/28/cell-build-from-scratch/|title=From chemicals to life: Scientists try to build cells from scratch|access-date=4 Dec 2019}}</ref> | ||
| − | |||
| − | |||
| − | |||
| − | == | + | 2017年,国际上大规模的人工合成活细胞建设研究合作正式启动,<ref>{{cite web|url=http://buildacell.io/|title=Build-a-Cell|access-date=4 Dec 2019}}</ref>随后若干国家也开展了国家间的合成细胞合作研究。这些全国性组织包括 FabriCell<ref>{{cite web|url=http://fabricell.org/|title=FabriCell|access-date=8 Dec 2019}}</ref>、 MaxSynBio <ref>{{cite web|url=https://www.maxsynbio.mpg.de/home/|title=MaxSynBio - Max Planck Research Network in Synthetic Biology|access-date=8 Dec 2019}}</ref>和 BaSyC。<ref>{{cite web|url=http://www.basyc.nl/|title=BaSyC|access-date=8 Dec 2019}}</ref>欧洲合成细胞的努力在2019年被统一为 SynCellEU 倡议。<ref>{{cite web|url=http://www.syntheticcell.eu/|title=SynCell EU|access-date=8 Dec 2019}}</ref> |
| − | |||
| − | |||
| − | + | ==自上而下创建一个最小活细胞的方法== | |
| + | 来自克莱格·凡特研究所 J. Craig Venter Institute的成员使用了一种自上而下的计算方法,将活生物体中的基因敲除至最小的一组基因。<ref name="gibson52" />2010年,研究小组利用人工合成中被认为是生命最低要求的 DNA,成功地创造了一个一株能复制的丝状支原体(支原体实验室) ,这种 DNA 被插入到一个基因组空白的细菌中。<ref name="gibson52" />人们希望,自上而下的生物合成过程将能够插入新的基因,这些基因将发挥有利的作用,例如产生氢气作为燃料或在大气中捕获过量的二氧化碳。<ref name="Beadau">{{cite book| veditors = Beadau MA | title=The ethics of protocells moral and social implications of creating life in the laboratory| year=2009| publisher=MIT Press| location=Cambridge, Mass.| isbn=978-0-262-51269-5| edition=[Online-Ausg.] | vauthors = Parke EC }}</ref>无数的调节、新陈代谢和信号网络没有被完全地表现,这些自上而下的方法对于理解基本的分子调控有局限性,因为宿主生物有一个复杂的和不完全确定的分子组成。<ref>{{cite journal | vauthors = Armstrong R | title = Designing with protocells: applications of a novel technical platform | journal = Life | volume = 4 | issue = 3 | pages = 457–490 | date = September 2014 | pmid = 25370381 | pmc = 4206855 | doi = 10.3390/life4030457 | doi-access = free }}</ref>2019年,支原体 Syn3.0细胞中所有通路的完整计算模型被发表,这是第一个完整的微生物活体硅模型。<ref>{{cite journal | vauthors = Breuer M, Earnest TM, Merryman C, Wise KS, Sun L, Lynott MR, Hutchison CA, Smith HO, Lapek JD, Gonzalez DJ, de Crécy-Lagard V, Haas D, Hanson AD, Labhsetwar P, Glass JI, Luthey-Schulten Z | display-authors = 6 | title = Essential metabolism for a minimal cell | journal = eLife | volume = 8 | date = January 2019 | pmid = 30657448 | pmc = 6609329 | doi = 10.7554/eLife.36842 }}</ref> | ||
| − | |||
| − | + | 大公司已经在生物领域投入了大量资金,比如与合成基因公司合作的埃克森美孚公司 ExxonMobil、克雷格·文特 Craig Venter的开发藻类燃料的生物合成公司<ref>{{cite journal | vauthors = Sheridan C | title = Big oil bucks for algae | journal = Nature Biotechnology | volume = 27 | issue = 9 | pages = 783 | date = September 2009 | pmid = 19741613 | doi = 10.1038/nbt0909-783 | s2cid = 205270805 }}</ref> | |
| − | |||
| − | + | 截至2016年,生殖支原体 Mycoplasma genitalium是唯一一种可以作为最小细胞工程起点的有机体,因为它拥有已知最小的可以在实验室条件下培育的基因组; 野生型品种有482个基因,去除正好100个被认为非必需的基因,就能培育出生长速度提高的可存活菌株。减少基因组的大肠杆菌是更有用的,而且已经开发出去除了15% 的基因组的可生存菌株。<ref name=":3">{{Cite journal|url=https://publications.europa.eu/en/publication-detail/-/publication/bfd7d06c-d3ae-11e5-a4b5-01aa75ed71a1/language-en|title=Opinion on synthetic biology II: Risk assessment methodologies and safety aspects |date=2016-02-12|language=en| author = EU Directorate-General for Health and Consumers |publisher=Publications Office |doi=10.2772/63529 }}</ref> | |
| − | + | 通过将一个完全合成的基因组引入到基因组上空出的宿主细胞中,即创造出了一种人工细胞。<ref name="gibson52">{{cite journal | vauthors = Gibson DG, Glass JI, Lartigue C, Noskov VN, Chuang RY, Algire MA, Benders GA, Montague MG, Ma L, Moodie MM, Merryman C, Vashee S, Krishnakumar R, Assad-Garcia N, Andrews-Pfannkoch C, Denisova EA, Young L, Qi ZQ, Segall-Shapiro TH, Calvey CH, Parmar PP, Hutchison CA, Smith HO, Venter JC | display-authors = 6 | title = Creation of a bacterial cell controlled by a chemically synthesized genome | journal = Science | volume = 329 | issue = 5987 | pages = 52–56 | date = July 2010 | pmid = 20488990 | doi = 10.1126/science.1190719 | s2cid = 7320517 | bibcode = 2010Sci...329...52G }}</ref>尽管由于宿主细胞的细胞质成分和细胞膜被保留下来而不完全是人工合成的,但该工程细胞处于合成基因组的控制之下,能够[[自复制]]。 | |
| − | |||
| + | ==医用人工细胞== | ||
| + | [[File:Standard and drug delivery artificial cells .png|thumb|350px|标准人工细胞(上)和药物传递人工细胞(下)。]] | ||
| − | |||
| − | == | + | ===历史=== |
| − | + | 20世纪60年代,Thomas Chang研制出了微胶囊,由于它们是由人造材料制成的细胞大小的隔室,后来他称之为“人工细胞”。<ref>{{cite journal | vauthors = Chang TM | title = SEMIPERMEABLE MICROCAPSULES | journal = Science | volume = 146 | issue = 3643 | pages = 524–525 | date = October 1964 | pmid = 14190240 | doi = 10.1126/science.146.3643.524 | s2cid = 40740134 | bibcode = 1964Sci...146..524C }}</ref>这些细胞由尼龙、火棉胶或交联蛋白质的超薄膜组成,其半透性使得小分子可以扩散进出细胞。这些细胞是微米大小,含有细胞,酶,血红蛋白,磁性材料,吸附剂和蛋白质。<ref name="Chang 2007" /> | |
| − | |||
| − | === | + | 后来人工细胞的尺寸从百微米到纳米不等,可以携带微生物、疫苗、基因、药物、激素和多肽。<ref name="Chang 2007" /> 人工细胞的第一次临床应用是以药用活性炭为包囊进行血液灌流。<ref name="Chang 1996">{{cite journal | vauthors = Chang TM | title=Editorial: past, present and future perspectives on the 40th anniversary of hemoglobin based red blood cell substitutes | journal=Artificial Cells Blood Substit Immobil Biotechnol | year=1996 | volume=24 | pages=ixxxvi }}</ref> |
| − | |||
| − | |||
| − | + | 在20世纪70年代,研究人员能够将酶、蛋白质和激素引入到可生物降解的微胶囊中,后来这种微胶囊在诸如莱希-尼亨氏症候群 Lesch–Nyhan syndrome之类的疾病中得到临床应用。<ref>{{cite journal | vauthors = Palmour RM, Goodyer P, Reade T, Chang TM | title = Microencapsulated xanthine oxidase as experimental therapy in Lesch-Nyhan disease | journal = Lancet | volume = 2 | issue = 8664 | pages = 687–688 | date = September 1989 | pmid = 2570944 | doi = 10.1016/s0140-6736(89)90939-2 | s2cid = 39716068 }}</ref>尽管Thomas Chang最初的研究集中在人工红细胞上,但直到20世纪90年代中期,才出现了可生物降解的人工红细胞。<ref>{{cite book | vauthors = Chang TM | title=Blood substitutes | year=1997 | publisher=Karger | location=Basel | isbn=978-3-8055-6584-4 }}</ref>1994年,生物细胞封装的人工细胞首次在临床上用于治疗糖尿病患者,<ref>{{cite journal | vauthors = Soon-Shiong P, Heintz RE, Merideth N, Yao QX, Yao Z, Zheng T, Murphy M, Moloney MK, Schmehl M, Harris M | display-authors = 6 | title = Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation | journal = Lancet | volume = 343 | issue = 8903 | pages = 950–951 | date = April 1994 | pmid = 7909011 | doi = 10.1016/S0140-6736(94)90067-1 | s2cid = 940319 }}</ref>此后,其他类型的细胞,如肝细胞、成体干细胞和基因工程细胞已被封装,并正在研究用于组织再生。<ref>{{cite journal | vauthors = Liu ZC, Chang TM | title = Coencapsulation of hepatocytes and bone marrow stem cells: in vitro conversion of ammonia and in vivo lowering of bilirubin in hyperbilirubemia Gunn rats | journal = The International Journal of Artificial Organs | volume = 26 | issue = 6 | pages = 491–497 | date = June 2003 | pmid = 12894754 | doi = 10.1177/039139880302600607 | s2cid = 12447199 }}</ref><ref>{{cite journal | vauthors = Aebischer P, Schluep M, Déglon N, Joseph JM, Hirt L, Heyd B, Goddard M, Hammang JP, Zurn AD, Kato AC, Regli F, Baetge EE | display-authors = 6 | title = Intrathecal delivery of CNTF using encapsulated genetically modified xenogeneic cells in amyotrophic lateral sclerosis patients | journal = Nature Medicine | volume = 2 | issue = 6 | pages = 696–699 | date = June 1996 | pmid = 8640564 | doi = 10.1038/nm0696-696 | s2cid = 8049662 }}</ref> | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
===材料=== | ===材料=== | ||
[[File:Artificial cell membranes.png|thumb|450px|典型的人工细胞膜。]] | [[File:Artificial cell membranes.png|thumb|450px|典型的人工细胞膜。]] | ||
| + | 用于人工细胞的膜可以由简单的聚合物、交联蛋白质、脂质膜或聚合物-脂质复合物制成。此外,膜可以被设计成表面蛋白如白蛋白、抗原、 Na/K-ATP 酶载体或孔隙如离子通道。常用的水凝胶聚合物如海藻酸钠、纤维素和热塑性聚合物如甲基丙烯酸羟乙酯-甲基丙烯酸甲酯(HEMA-MMA)、聚丙烯腈-聚氯乙烯(PAN-PVC) ,以及上述各种聚合物的变异。<ref name=Prakash/>所用的材料决定了细胞膜的渗透性,对于聚合物来说,这取决于截留分子量(MWCO)。<ref name=Prakash/>MWCO 是可以自由通过孔隙的分子的最大分子量,对于确定营养物质、废物和其他临界分子的充分扩散很重要。亲水性聚合物具有生物相容性的潜力,可以制成多种形式,包括聚合物胶束、溶胶-凝胶混合物、物理共混物以及交联粒子和纳米粒子。<ref name=Prakash/>特别令人感兴趣的是对pH值或温度变化有反应的刺激性聚合物,用于靶向传递。由于 pH 值和温度的不同,这些聚合物可以通过宏观注射和凝固或原位凝胶以液体形式给药。纳米颗粒和脂质体制剂也常用于材料的包封和递送。脂质体的一个主要优点是它们能够与细胞膜和细胞器膜融合。 | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| + | ===制备=== | ||
人工细胞的制备和包封已经发展出许多不同的方法。通常,如纳米粒子、聚合物或脂质体等小泡被合成。乳化液通常是通过高压设备,如高压匀浆器或微射流机来制造的。下面将介绍硝化纤维素的两种微胶囊化方法. | 人工细胞的制备和包封已经发展出许多不同的方法。通常,如纳米粒子、聚合物或脂质体等小泡被合成。乳化液通常是通过高压设备,如高压匀浆器或微射流机来制造的。下面将介绍硝化纤维素的两种微胶囊化方法. | ||
| − | ====High-pressure homogenization==== | + | ====高压均质化 High-pressure homogenization==== |
| − | |||
| − | |||
在高压匀浆器中,油/液悬浮液中的两种液体在很高的压力下通过一个小孔。这一过程划分了产品,并允许产生极细的颗粒,小到1nm。 | 在高压匀浆器中,油/液悬浮液中的两种液体在很高的压力下通过一个小孔。这一过程划分了产品,并允许产生极细的颗粒,小到1nm。 | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | ==== | + | ====微射流处理 Microfluidization==== |
| − | + | 微射流技术使用专利的微射流机获得更多的均质悬浮液,可以产生比匀浆器更小的颗粒。匀浆器首先被用来制造粗颗粒悬浮液,然后在高压下被泵入微射流机。然后将流体分成两股流体,这两股流体将在相互作用室中以非常高的速度反应,直到得到所需的粒子尺寸。<ref>{{cite journal | vauthors = Vivier A, Vuillemard JC, Ackermann HW, Poncelet D | title = Large-scale blood substitute production using a microfluidizer | journal = Biomaterials, Artificial Cells, and Immobilization Biotechnology | volume = 20 | issue = 2-4 | pages = 377–397 | year = 1992 | pmid = 1391454 | doi = 10.3109/10731199209119658 }}</ref>这种技术允许大规模生产磷脂脂质体和后续材料纳米包埋。 | |
| + | ====点滴法 Drop method==== | ||
在这种方法中,细胞溶液滴入硝酸纤维素的火棉胶溶液中。当液滴通过火棉胶时,由于火棉胶的界面聚合特性,它被涂上了一层膜。细胞随后沉积在石蜡中,细胞膜凝固,最后悬浮在盐溶液中。点滴法用于制造大型人工细胞,包裹生物细胞、干细胞和基因工程干细胞。 | 在这种方法中,细胞溶液滴入硝酸纤维素的火棉胶溶液中。当液滴通过火棉胶时,由于火棉胶的界面聚合特性,它被涂上了一层膜。细胞随后沉积在石蜡中,细胞膜凝固,最后悬浮在盐溶液中。点滴法用于制造大型人工细胞,包裹生物细胞、干细胞和基因工程干细胞。 | ||
| − | |||
| − | |||
| + | ====乳化法 Emulsion method==== | ||
| + | 乳化法的不同之处在于,要被包裹的材料通常较小,被放置在反应室的底部,在反应室的顶部加入火棉胶并离心,或以其他方式搅拌以便形成乳化液。然后将胶囊材料分散并悬浮在盐溶液中。 | ||
| − | |||
===材料=== | ===材料=== | ||
| − | ==== | + | ====药物的释放和运送==== |
| − | |||
| − | |||
| − | |||
| − | |||
用于药物运送的人工细胞与其他人工细胞不同,是由于其内容物要扩散出细胞膜,或者被宿主靶细胞吞噬和消化。通常使用的是亚微米,脂质膜人工细胞,可称为纳米胶囊,纳米粒子,聚合体,或其他变体。 | 用于药物运送的人工细胞与其他人工细胞不同,是由于其内容物要扩散出细胞膜,或者被宿主靶细胞吞噬和消化。通常使用的是亚微米,脂质膜人工细胞,可称为纳米胶囊,纳米粒子,聚合体,或其他变体。 | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | ====酶疗法 Enzyme therapy==== | |
| + | 目前正在积极研究酶疗法,用于治疗有某种酶过度表达、表达不足、有缺陷或根本不存在等表现的遗传代谢性疾病。在有缺陷的酶的表达不足或表达不足的情况下,活性形式的该种酶将被引入人体以弥补缺陷。另一方面,酶的过度表达可以通过引入竞争性的非功能性酶来抵消,也就是说,一种将底物代谢为非活性产物的酶。当置于人工细胞中时,酶可以比游离酶更长时间地发挥作用,<ref name= 'Chang 2007' />并且可以通过聚合物接合进一步优化。<ref>Park et al. 1981</ref> | ||
| − | + | 人工细胞包裹法研究的第一种酶是治疗小鼠淋巴肉瘤的天冬酰胺酶。<ref>{{cite journal | vauthors = Chang TM | title = The in vivo effects of semipermeable microcapsules containing L-asparaginase on 6C3HED lymphosarcoma | journal = Nature | volume = 229 | issue = 5280 | pages = 117–118 | date = January 1971 | pmid = 4923094 | doi = 10.1038/229117a0 | s2cid = 4261902 | bibcode = 1971Natur.229..117C }}</ref>这种治疗延缓了肿瘤的发生和生长。<ref>{{cite journal | vauthors = Yu B, Chang TM | title = Effects of long-term oral administration of polymeric microcapsules containing tyrosinase on maintaining decreased systemic tyrosine levels in rats | journal = Journal of Pharmaceutical Sciences | volume = 93 | issue = 4 | pages = 831–837 | date = April 2004 | pmid = 14999721 | doi = 10.1002/jps.10593 }}</ref>这些初步的发现导致了在酪氨酸依赖性黑色素瘤中使用人工细胞进行酶传递的进一步研究。这些肿瘤的生长比正常细胞更依赖于酪氨酸,研究表明,降低小鼠全身酪氨酸水平可以抑制黑色素瘤的生长。<ref>{{cite journal | vauthors = Meadows GG, Pierson HF, Abdallah RM, Desai PR | title = Dietary influence of tyrosine and phenylalanine on the response of B16 melanoma to carbidopa-levodopa methyl ester chemotherapy | journal = Cancer Research | volume = 42 | issue = 8 | pages = 3056–3063 | date = August 1982 | pmid = 7093952 }}</ref>利用人工细胞输送酪氨酸酶和消化酪氨酸的酶,可以提高酶的稳定性,并且能有效地去除酪氨酸,而不会产生与饮食中酪氨酸恶化有关的严重副作用。<ref>{{cite journal | vauthors = Chang TM | title = Artificial cell bioencapsulation in macro, micro, nano, and molecular dimensions: keynote lecture | journal = Artificial Cells, Blood Substitutes, and Immobilization Biotechnology | volume = 32 | issue = 1 | pages = 1–23 | date = February 2004 | pmid = 15027798 | doi = 10.1081/bio-120028665 | s2cid = 37799530 }}</ref> | |
| − | |||
| − | |||
| − | + | 在某些癌症中,人工细胞酶疗法对于激活诸如异环磷酰胺之类的前药也很有意义。将细胞色素 p450酶包裹在人工细胞中,将其转化为活性药物,可以特制地在胰腺癌中积累,或将人工细胞移植到肿瘤部位附近。在这里,被激活的异环磷酰胺的局部浓度将远远高于身体其他部位,从而防止全身中毒。<ref name='Lohr'>{{cite journal | vauthors = Löhr M, Hummel F, Faulmann G, Ringel J, Saller R, Hain J, Günzburg WH, Salmons B | display-authors = 6 | title = Microencapsulated, CYP2B1-transfected cells activating ifosfamide at the site of the tumor: the magic bullets of the 21st century | journal = Cancer Chemotherapy and Pharmacology | volume = 49 | issue = Suppl 1 | pages = S21-S24 | date = May 2002 | pmid = 12042985 | doi = 10.1007/s00280-002-0448-0 | s2cid = 10329480 }}</ref>这种治疗在动物身上取得了成功,<ref>{{cite journal | vauthors = Kröger JC, Benz S, Hoffmeyer A, Bago Z, Bergmeister H, Günzburg WH, Karle P, Klöppel G, Losert U, Müller P, Nizze H, Obermaier R, Probst A, Renner M, Saller R, Salmons B, Schwendenwein I, von Rombs K, Wiessner R, Wagner T, Hauenstein K, Löhr M | display-authors = 6 | title = Intra-arterial instillation of microencapsulated, Ifosfamide-activating cells in the pig pancreas for chemotherapeutic targeting | journal = Pancreatology | volume = 3 | issue = 1 | pages = 55–63 | year = 1999 | pmid = 12649565 | doi = 10.1159/000069147 | s2cid = 23711385 }}</ref>在 I/II 期临床试验中,晚期胰腺癌患者的生存率中位数增加了一倍,一年生存率增加了两倍。<ref name=Lohr/> | |
| − | + | ====基因疗法 Gene therapy==== | |
| + | 在基因疾病的治疗中,基因疗法旨在插入、改变或移除受影响个体细胞内的基因。该技术严重依赖于病毒载体,这引起了对插入突变和系统免疫反应的关注,它们在临床试验中导致了人类死亡<ref>{{cite journal | vauthors = Carmen IH | title = A death in the laboratory: the politics of the Gelsinger aftermath | journal = Molecular Therapy | volume = 3 | issue = 4 | pages = 425–428 | date = April 2001 | pmid = 11319902 | doi = 10.1006/mthe.2001.0305 }}</ref><ref>{{cite journal | vauthors = Raper SE, Chirmule N, Lee FS, Wivel NA, Bagg A, Gao GP, Wilson JM, Batshaw ML | display-authors = 6 | title = Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer | journal = Molecular Genetics and Metabolism | volume = 80 | issue = 1-2 | pages = 148–158 | date = 1 September 2003 | pmid = 14567964 | doi = 10.1016/j.ymgme.2003.08.016 }}</ref>和白血病的发展<ref>{{cite journal | vauthors = Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P, Selz F, Hue C, Certain S, Casanova JL, Bousso P, Deist FL, Fischer A | display-authors = 6 | title = Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease | journal = Science | volume = 288 | issue = 5466 | pages = 669–672 | date = April 2000 | pmid = 10784449 | doi = 10.1126/science.288.5466.669 | bibcode = 2000Sci...288..669C }}</ref><ref>{{cite journal | vauthors = Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack MP, Wulffraat N, Leboulch P, Lim A, Osborne CS, Pawliuk R, Morillon E, Sorensen R, Forster A, Fraser P, Cohen JI, de Saint Basile G, Alexander I, Wintergerst U, Frebourg T, Aurias A, Stoppa-Lyonnet D, Romana S, Radford-Weiss I, Gross F, Valensi F, Delabesse E, Macintyre E, Sigaux F, Soulier J, Leiva LE, Wissler M, Prinz C, Rabbitts TH, Le Deist F, Fischer A, Cavazzana-Calvo M | display-authors = 6 | title = LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1 | journal = Science | volume = 302 | issue = 5644 | pages = 415–419 | date = October 2003 | pmid = 14564000 | doi = 10.1126/science.1088547 | s2cid = 9100335 | bibcode = 2003Sci...302..415H }}</ref>。利用裸DNA或质粒DNA作为载体载体本身的传递系统,避免了载体的需要,同时也遇到了系统传递效率低、组织靶向性差等问题。<ref name=Prakash/> | ||
| − | |||
| + | 人工细胞被认为是一种非病毒载体,将转基因非自体细胞包裹起来并植入体内以表达重组蛋白。<ref>{{cite journal | vauthors = Chang PL, Van Raamsdonk JM, Hortelano G, Barsoum SC, MacDonald NC, Stockley TL | title = The in vivo delivery of heterologous proteins by microencapsulated recombinant cells | journal = Trends in Biotechnology | volume = 17 | issue = 2 | pages = 78–83 | date = February 1999 | pmid = 10087608 | doi = 10.1016/S0167-7799(98)01250-5 }}</ref>通过传递含有小鼠生长激素的人工细胞,挽救了突变小鼠的生长迟缓,这种免疫分离已经被证明是有效的。<ref>{{cite journal | vauthors = al-Hendy A, Hortelano G, Tannenbaum GS, Chang PL | title = Correction of the growth defect in dwarf mice with nonautologous microencapsulated myoblasts--an alternate approach to somatic gene therapy | journal = Human Gene Therapy | volume = 6 | issue = 2 | pages = 165–175 | date = February 1995 | pmid = 7734517 | doi = 10.1089/hum.1995.6.2-165 }}</ref>一些治疗胰腺癌、脊髓侧索硬化症和疼痛控制的方案已经进入人体临床试验阶段。<ref name= 'Prakash'/> | ||
| − | |||
| − | ====Hemoperfusion==== | + | ====血液灌流 Hemoperfusion==== |
| − | + | 临床上第一次使用人工细胞的方法是用活性炭包裹血液灌流。<ref name="Chang 1996"/>药用活性炭具有吸附许多大分子的能力,长期以来因其在意外中毒或过量服用中能够去除血液中的有毒物质而闻名。然而,通过直接使用木炭进行灌注是有毒的,因为它会导致血栓和血细胞损伤,随后被血小板清除。<ref>{{cite journal | vauthors = Dunea G, Kolff WJ | title = CLINICAL EXPERIENCE WITH THE YATZIDIS CHARCOAL ARTIFICIAL KIDNEY | journal = Transactions - American Society for Artificial Internal Organs | volume = 11 | pages = 178–182 | year = 1965 | pmid = 14329080 | doi = 10.1097/00002480-196504000-00035 }}</ref>人工细胞允许毒素扩散到细胞中,同时将危险物质保存在超薄的膜内。<ref name= 'Chang 1996' /> | |
| − | |||
| + | 与血液透析相比,人工细胞血液灌流被认为是一种成本更低、效率更高的解毒方法。<ref name="Chang 2007"/>在血液灌流中,成千上万的吸附性人工细胞通过在病人血液灌流的两端使用两个筛子被保存在一个小容器内。随着血液循环,毒素或药物扩散进入细胞,并被吸收材料所保留。人工细胞的细胞膜比透析用细胞的细胞膜要薄得多,细胞膜的小体积意味着细胞膜的表面积很大。这意味着一部分细胞理论上的质量传递比整个人工肾脏机器的质量传递高百倍。<ref name='Chang 2007' />该装置已被确立为治疗意外或自杀性中毒患者的常规临床方法,但也被引入治疗肝功能衰竭和肾功能衰竭,以实现这些器官的部分功能。<ref name= 'Chang 2007' />人工细胞血液灌流也被提议用于免疫吸附,通过在人工细胞表面粘附免疫吸附材料,如白蛋白,可将抗体从体内除去。这一原理已被应用于从骨髓移植患者血浆中去除血型抗体,<ref>{{cite journal | vauthors = Bensinger WI, Buckner CD, Clift RA | title = Whole blood immunoadsorption of anti-A or anti-B antibodies | journal = Vox Sanguinis | volume = 48 | issue = 6 | pages = 357–361 | year = 1985 | pmid = 3892895 | doi = 10.1111/j.1423-0410.1985.tb00196.x | s2cid = 12777645 }}</ref>以及通过单克隆抗体去除低密度脂蛋白治疗高胆固醇血症。<ref>{{cite journal | vauthors = Yang L, Cheng Y, Yan WR, Yu YT | title = Extracorporeal whole blood immunoadsorption of autoimmune myasthenia gravis by cellulose tryptophan adsorbent | journal = Artificial Cells, Blood Substitutes, and Immobilization Biotechnology | volume = 32 | issue = 4 | pages = 519–528 | year = 2004 | pmid = 15974179 | doi = 10.1081/bio-200039610 | s2cid = 7269229 }}</ref>血液灌流在血液透析制造业薄弱的国家尤其有用,因为那里的血液透析设备往往更便宜,而且用于肾衰竭患者。 | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
====微囊细胞==== | ====微囊细胞==== | ||
| 第189行: | 第125行: | ||
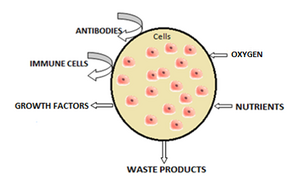
[[File:Cell capsule schematic.png|thumb|300px|Schematic representation of encapsulated cells within artificial membrane.人工膜内微囊细胞的原理图表示。]] | [[File:Cell capsule schematic.png|thumb|300px|Schematic representation of encapsulated cells within artificial membrane.人工膜内微囊细胞的原理图表示。]] | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | 最常用的制备人工细胞的方法是细胞包裹。微囊细胞通常由液体细胞悬浮液中产生控制尺寸的液滴获取,然后快速凝固或凝胶化以提供额外的稳定性。稳定化可以通过温度的变化或材料的交联来实现。细胞的微环境在包裹后发生变化,且通常从单分子层到悬浮在聚合物膜内的聚合物支架上。这种技术的缺点是微囊细胞会降低其生存能力以及增殖和分化能力。<ref>{{cite book | veditors = Wolff JA | vauthors = Chang PL | date = 1994 | chapter = Calcium phosphate-mediated DNA transfection | pages = 157–179 | title = Gene Therapeutics | location = Boston | publisher = Birkhauser | isbn = 978-1-4684-6822-9 | doi = 10.1007/978-1-4684-6822-9_9 }}</ref>此外,在微囊中待一段时间后,细胞会形成团簇,抑制氧气和代谢废物的交换,<ref>{{cite journal | vauthors = Ponce S, Orive G, Gascón AR, Hernández RM, Pedraz JL | title = Microcapsules prepared with different biomaterials to immobilize GDNF secreting 3T3 fibroblasts | journal = International Journal of Pharmaceutics | volume = 293 | issue = 1-2 | pages = 1–10 | date = April 2005 | pmid = 15778039 | doi = 10.1016/j.ijpharm.2004.10.028 }}</ref>导致细胞凋亡和坏死,从而限制细胞的功效,激活宿主的免疫系统。人工细胞已经成功地移植了一些细胞,包括治疗糖尿病的胰岛、<ref>{{cite journal | vauthors = Kizilel S, Garfinkel M, Opara E | title = The bioartificial pancreas: progress and challenges | journal = Diabetes Technology & Therapeutics | volume = 7 | issue = 6 | pages = 968–985 | date = December 2005 | pmid = 16386103 | doi = 10.1089/dia.2005.7.968 }}</ref>甲状旁腺细胞和肾上腺皮质细胞。 | |
| − | |||
| − | |||
| − | |||
| + | ====微囊肝细胞 Encapsulated hepatocytes==== | ||
| + | 器官捐献者的短缺使人工细胞成为治疗肝衰竭替代疗法的关键。人工细胞用于肝细胞移植已被证实在动物肝病模型和生物人工肝装置中提供肝功能的可行性和有效性。<ref name="Dixit">{{cite journal | vauthors = Dixit V, Gitnick G | title = The bioartificial liver: state-of-the-art | journal = The European Journal of Surgery. Supplement. | volume = 164 | issue = 582 | pages = 71–76 | date = 27 November 2003 | pmid = 10029369 | doi = 10.1080/11024159850191481 | name-list-style = vanc }}</ref>实验中,肝细胞附着在微载体表面,<ref name="pmid2426782">{{cite journal | vauthors = Demetriou AA, Whiting JF, Feldman D, Levenson SM, Chowdhury NR, Moscioni AD, Kram M, Chowdhury JR | display-authors = 6 | title = Replacement of liver function in rats by transplantation of microcarrier-attached hepatocytes | journal = Science | volume = 233 | issue = 4769 | pages = 1190–1192 | date = September 1986 | pmid = 2426782 | doi = 10.1126/science.2426782 | bibcode = 1986Sci...233.1190D }}</ref>然后进化成包裹在由聚赖氨酸外皮覆盖的海藻酸钠微滴中的三维基质中的肝细胞。这种给药方法的一个关键优势是在治疗期间避免了免疫抑制治疗。微囊肝细胞已被提议用于生物人工肝。该装置由一个嵌有分离肝细胞的圆柱形腔室组成,病人的血浆通过这个腔室在一种血液灌流中循环。由于微囊具有很高的比表面积和体积比,它们为基质扩散提供了较大的表面积,并且可以容纳大量的肝细胞。对诱导性肝衰竭小鼠的治疗显示存活率显著提高。<ref name="Dixit" />人工肝系统仍处于早期发展阶段,但对于等待器官移植或者病人自身的肝脏重新充分恢复正常功能的病人来说,显示出了潜力。迄今为止,应用人工肝系统和肝细胞移植治疗终末期肝病的临床试验表明,健康指标有所改善,但还没有提高存活率。<ref>{{cite journal | vauthors = Sgroi A, Serre-Beinier V, Morel P, Bühler L | title = What clinical alternatives to whole liver transplantation? Current status of artificial devices and hepatocyte transplantation | journal = Transplantation | volume = 87 | issue = 4 | pages = 457–466 | date = February 2009 | pmid = 19307780 | doi = 10.1097/TP.0b013e3181963ad3 }}</ref>移植后人工肝细胞的短寿命和聚集是目前肝移植面临的主要障碍。<ref>{{cite journal | vauthors = Liu ZC, Chang TM | title = Increased viability of transplanted hepatocytes when hepatocytes are co-encapsulated with bone marrow stem cells using a novel method | journal = Artificial Cells, Blood Substitutes, and Immobilization Biotechnology | volume = 30 | issue = 2 | pages = 99–112 | date = March 2002 | pmid = 12027231 | doi = 10.1081/bio-120003191 | s2cid = 26667880 }}</ref>与干细胞一同包裹的肝细胞在培养和植入后显示出更高的存活率,单独植入人工干细胞也可以促成肝的再生。<ref>{{cite book| veditors = Pedraz JL, Orive G |title=Therapeutic applications of cell microencapsulation|year=2010|publisher=Springer Science+Business Media|location=New York|isbn=978-1-4419-5785-6|edition=Online-Ausg.}}</ref>因此,在再生医学中利用干细胞封装的研究引起了人们的兴趣。 | ||
| − | |||
| − | ==== | + | ====微囊细菌细胞 Encapsulated bacterial cells==== |
| − | + | 口服活菌群,目前正用于治疗调节肠道菌群,<ref>{{cite journal | vauthors = Mattila-Sandholm T, Blum S, Collins JK, Crittenden R, De Vos W, Dunne C, Fondén R, Grenov G, Isolauri E, Kiely B, Marteau P, Morelli L, Ouwehand A, Reniero R, Saarela M, Salminen S, Saxelin M, Schiffrin E, Shanahan F, Vaughan E, von Wright A | display-authors = 6 |title=Probiotics: towards demonstrating efficacy|journal=Trends in Food Science & Technology|date=1 December 1999 |volume=10|issue=12|pages=393–399|doi=10.1016/S0924-2244(00)00029-7 }}</ref>预防腹泻疾病,<ref>{{cite journal | vauthors = Huang JS, Bousvaros A, Lee JW, Diaz A, Davidson EJ | title = Efficacy of probiotic use in acute diarrhea in children: a meta-analysis | journal = Digestive Diseases and Sciences | volume = 47 | issue = 11 | pages = 2625–2634 | date = November 2002 | pmid = 12452406 | doi = 10.1023/A:1020501202369 | s2cid = 207559325 }}</ref>治疗幽门螺杆菌感染,特应性炎症,<ref>{{cite journal | vauthors = Isolauri E, Arvola T, Sütas Y, Moilanen E, Salminen S | title = Probiotics in the management of atopic eczema | journal = Clinical and Experimental Allergy | volume = 30 | issue = 11 | pages = 1604–1610 | date = November 2000 | pmid = 11069570 | doi = 10.1046/j.1365-2222.2000.00943.x | s2cid = 13524021 }}</ref>乳糖不耐受<ref>{{cite journal | vauthors = Lin MY, Yen CL, Chen SH | title = Management of lactose maldigestion by consuming milk containing lactobacilli | journal = Digestive Diseases and Sciences | volume = 43 | issue = 1 | pages = 133–137 | date = January 1998 | pmid = 9508514 | doi = 10.1023/A:1018840507952 | s2cid = 22890925 }}</ref>和免疫调节<ref>{{cite journal| vauthors = Gill HS |title=Stimulation of the Immune System by Lactic Cultures|journal=International Dairy Journal|date=1 May 1998 |volume=8|issue=5–6|pages=535–544|doi=10.1016/S0958-6946(98)00074-0}}</ref>等症病。虽然其作用机制尚未得到充分理解,但被认为其具有两个主要作用。首先是营养效应,即细菌与产毒细菌竞争。第二是卫生效应,即刺激抵抗定殖和免疫反应。<ref name="Prakash" />由于细菌培养物往往是免疫系统的目标,通常会在口服时被破坏,口服给药的方式便成了困难。人工细胞通过向体内提供拟态,以及其可选择的或长期的释放,来提高进入胃肠道系统的细菌的生存能力,从而帮助解决这些问题。<ref name="Prakash">{{cite book| vauthors = Prakash S |title=Artificial cells, cell engineering and therapy.|year=2007|publisher=Woodhead Publishing Limited|location=Boca Raton, Fl|isbn=978-1-84569-036-6}}</ref>此外,还可以设计活细菌包囊,使小分子(包括多肽)扩散到体内,以用于治疗。<ref name="Prakash" />醋酸纤维素和海藻酸盐的变体等已被证明,可以成功用于细菌传递的膜。<ref name="Prakash" />微囊细菌细胞的其他用途还包括保护细菌细胞免受结核分枝杆菌的攻击,<ref>{{cite journal | vauthors = Aldwell FE, Tucker IG, de Lisle GW, Buddle BM | title = Oral delivery of Mycobacterium bovis BCG in a lipid formulation induces resistance to pulmonary tuberculosis in mice | journal = Infection and Immunity | volume = 71 | issue = 1 | pages = 101–108 | date = January 2003 | pmid = 12496154 | pmc = 143408 | doi = 10.1128/IAI.71.1.101-108.2003 }}</ref>以及促进免疫系统产生Ig分泌细胞。<ref>{{cite journal | vauthors = Park JH, Um JI, Lee BJ, Goh JS, Park SY, Kim WS, Kim PH | title = Encapsulated Bifidobacterium bifidum potentiates intestinal IgA production | journal = Cellular Immunology | volume = 219 | issue = 1 | pages = 22–27 | date = September 2002 | pmid = 12473264 | doi = 10.1016/S0008-8749(02)00579-8 }}</ref>该技术受到系统性感染、不良代谢活动和基因转移风险的限制。<ref name="Prakash" />然而,更大的挑战仍然是如何将足够多的有生命的细菌运送到目标部位。<ref name="Prakash" /> | |
| − | |||
| − | |||
| + | ====人工血细胞作为氧载体 Artificial blood cells as oxygen carriers==== | ||
| + | 人工血细胞作为氧载体被用作一种红细胞替代品,尽管它们缺乏红细胞的其他成分。它们由一种合成的聚合体或围绕着纯化的动物、人或重组的血红蛋白的人工膜组成。<ref>{{cite journal | vauthors = Kim HW, Greenburg AG | title = Artificial oxygen carriers as red blood cell substitutes: a selected review and current status | journal = Artificial Organs | volume = 28 | issue = 9 | pages = 813–828 | date = September 2004 | pmid = 15320945 | doi = 10.1111/j.1525-1594.2004.07345.x }}</ref>总的来说,血红蛋白的输送仍然是一个挑战,因为它在没有任何修饰的情况下输送时是剧毒的。在一些临床试验中,人们观察到血管升压效应。<ref>{{cite book | vauthors = Nelson DJ | date = 1998 | chapter = Blood and HemAssistTM (DCLHb): Potentially a complementary therapeutic team | title = Blood Substitutes: Principles, Methods, Products and Clinical Trials | veditors = Chang TM | volume = 2 | publisher = Karger | location = Basel | pages = 39–57 }}</ref><ref>{{cite journal | vauthors = Burhop KE, Estep TE | date = 2001 | title = Hemoglobin induced myocardial lesions | journal = Artificial Cells, Blood Substitutes, and Biotechnology | volume = 29 | issue = 2 | pages = 101–106 | doi = 10.1080/10731190108951271 | pmc = 3555357 }}</ref> | ||
| − | |||
| − | ====Artificial | + | ====人工红细胞 Artificial red blood cells==== |
| − | |||
| − | + | 在1980年代的艾滋病恐慌之后,人们对使用人工红细胞制造血液产生了研究兴趣。除了避开疾病传播的可能性之外,人工红细胞也是人们所需要的,因为它们可以消除与同种异体输血相关的缺点,如血型鉴定、免疫反应及其短暂的42天储存期。血红蛋白替代物可以存放在室温下,而不是在冷藏条件下存放一年以上。<ref name= 'Chang 2007' />人们已经尝试开发出一种完整工作的红细胞,它不仅包含碳酸,而且包含氧载体和与细胞相关的酶。第一次尝试是在1957年,用超薄聚合物膜<ref>{{cite journal|title=30th Anniversary in Artificial Red Blood Cell Research|journal=Artificial Cells, Blood Substitutes and Biotechnology|date=1 January 1988 |volume=16|issue=1–3|pages=1–9|doi=10.3109/10731198809132551}}</ref>代替红细胞膜,然后通过脂质膜<ref>{{cite journal | vauthors = Djordjevich L, Miller IF | title = Synthetic erythrocytes from lipid encapsulated hemoglobin | journal = Experimental Hematology | volume = 8 | issue = 5 | pages = 584–592 | date = May 1980 | pmid = 7461058 }}</ref>和最近的可生物降解聚合物膜进行包封。<ref name= 'Chang 2007' />包括脂类和相关蛋白在内的生物红细胞膜还可以通过绕过巨噬细胞摄取和系统清除来包裹纳米颗粒和增加体内滞留时间。<ref>{{cite journal | vauthors = Hu CM, Zhang L, Aryal S, Cheung C, Fang RH, Zhang L | title = Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 108 | issue = 27 | pages = 10980–10985 | date = July 2011 | pmid = 21690347 | pmc = 3131364 | doi = 10.1073/pnas.1106634108 | doi-access = free | bibcode = 2011PNAS..10810980H }}</ref> | |
| − | |||
| − | == | + | ====人工白细胞聚合体 Artificial leuko-polymersomes ==== |
| − | + | 人工白细胞聚合体是一种具有白细胞粘附特性的高分子囊泡。<ref>{{cite journal | vauthors = Hammer DA, Robbins GP, Haun JB, Lin JJ, Qi W, Smith LA, Ghoroghchian PP, Therien MJ, Bates FS | display-authors = 6 | title = Leuko-polymersomes | journal = Faraday Discussions | volume = 139 | pages = 129–41; discussion 213–28, 419–20 | date = 1 January 2008 | pmid = 19048993 | pmc = 2714229 | doi = 10.1039/B717821B | bibcode = 2008FaDi..139..129H }}</ref>高分子囊泡是由双层薄片组成的囊泡,可以包裹许多活性分子,如药物或酶。通过将白细胞的粘附特性添加到细胞膜上,白细胞可以减慢速度,或者在快速流动的循环系统内沿着上皮细胞壁滚动。 | |
| − | |||
| + | ==非常规类型的人工细胞== | ||
| + | ===电子人工细胞 Electronic artificial cell=== | ||
电子人工细胞的概念在2004年至2015年由约翰 · 麦卡斯基尔协调的一系列欧盟项目中得到扩展。 | 电子人工细胞的概念在2004年至2015年由约翰 · 麦卡斯基尔协调的一系列欧盟项目中得到扩展。 | ||
| − | |||
| + | 2004年至2008年,欧洲联盟委员会赞助制定了可编程人工细胞进化方案<ref name = "PACE">{{cite web | title = Programmable Artificial Cell Evolution" (PACE) | url = http://www.istpace.org/Web_Final_Report/the_pace_report/index.html?View=default | publisher = PACE Consortium }}</ref>,其目标是为最终融入信息系统奠定基础,创建”由简单的有机和无机物质构成的微观自组织、自我复制和可进化的自主实体,这些物质可通过遗传程序进行编程,以履行特定功能”<ref name = "PACE" />。PACE 项目开发了第一台欧米茄机器,这是一种人工细胞的微流体生命支持系统,可以补充化学缺失的功能(最初由Norman Packard、Steen Rasmussen、Mark Beadau和John McCaskill基尔提出)。最终目标是在复杂的微型可编程环境中实现可进化的混合细胞。欧米茄机器的功能可以逐步被移除,这对人工细胞化学提出了一系列可解决的进化挑战。该项目实现了人工细胞三个核心功能(遗传子系统、包容系统和新陈代谢系统)成对的化学整合,并为包容和基因扩增的整合创造了新的空间分辨可编程微流体环境。<ref name = "PACE" />这个项目导致了欧洲生物技术中心的建立。<ref>{{cite web | title = European center for living technology | url = http://www.ecltech.org/ecltech_j/ | archive-url = https://web.archive.org/web/20111214010917/http://www.ecltech.org/ecltech_j/ | url-status = dead | archive-date = 2011-12-14 | publisher = European Center for Living Technology }}</ref> | ||
| − | |||
| − | + | 在这项研究之后,2007年,John McCaskill基尔提议集中研究一种电子补充型人工电池,称为电子化学电池。关键的想法是在一个二维薄膜中,使用一个大规模并行处理机阵列的电极与局部专用电子电路耦合,以补充新出现的化学细胞功能。定义电极切换和传感电路的局部电子信息可以作为电子基因组,补充新协议中的分子序列信息。与欧洲联盟委员会的一项研究提案取得了成功,与计算机设备行动伙伴关系联盟部分重叠的一个国际科学家小组开始了2008-2012年电子化学细胞项目的工作。除其他外,该项目表明,电子控制的特定序列的局部转移可以用作未来人工细胞遗传增殖的人工空间控制系统,并且新陈代谢的核心过程可以通过适当的涂层电极阵列传递。 | |
| − | |||
| − | |||
| − | |||
| 第255行: | 第163行: | ||
| − | ===Jeewanu=== | + | ===杰瓦努 Jeewanu=== |
| − | + | 杰瓦努原始细胞是具有细胞样结构的合成化学粒子,似乎具有一定的功能活性。<ref name="Grote 2011">{{cite journal | vauthors = Grote M | title = Jeewanu, or the 'particles of life'. The approach of Krishna Bahadur in 20th century origin of life research | journal = Journal of Biosciences | volume = 36 | issue = 4 | pages = 563–570 | date = September 2011 | pmid = 21857103 | doi = 10.1007/s12038-011-9087-0 | url = http://www.ias.ac.in/jbiosci/grote_3677.pdf | url-status = dead | s2cid = 19551399 | archive-url = https://web.archive.org/web/20140323225723/http://www.ias.ac.in/jbiosci/grote_3677.pdf | archive-date = 2014-03-23 }}</ref>1963年首次在阳光下由简单矿物质和基本有机物合成,据报道它仍然具有一些新陈代谢能力,包括半透膜、氨基酸、磷脂、碳水化合物和类 RNA分子。然而,杰瓦努的性质和属性仍有待澄清。<ref name="Grote 2011" /><ref name="Gupta 2013">{{cite journal |title=Histochemical localisation of RNA-like material in photochemically formed self-sustaining, abiogenic supramolecular assemblies 'Jeewanu' |journal=Int. Res. J. Of Science & Engineering |date=2013 | vauthors = Gupta VK, Rai RK |volume=1 |issue=1 |pages=1–4 |issn=2322-0015 }}</ref> However, the nature and properties of the Jeewanu remains to be clarified.<ref name="Grote 2011" /><ref name="Gupta 2013" /><ref name="NASA 1967">{{cite journal |first1=Linda D. |last1=Caren |first2=Cyril |last2=Ponnamperuma |year=1967 |url=https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/19670026284.pdf |title=A review of some experiments on the synthesis of 'Jeewanu' |journal=NASA Technical Memorandum X-1439 }}</ref> | |
| − | |||
| − | |||
| − | |||
== See also == | == See also == | ||
| − | * [[ | + | * [[原始细胞]] |
| − | * [[ | + | * [[合成生物学]] |
| − | * [[ | + | * [[人工生命]] |
| − | * [[ | + | * [[靶向给药]] |
| − | * [[ | + | * [[呼吸细胞]] |
| − | * [[ | + | * [[化学药品]] |
| − | * [[ | + | * [[杰瓦努]] |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | == | + | == 参考文献 == |
{{Reflist}} | {{Reflist}} | ||
| − | == | + | |
| + | == 外部链接 == | ||
*[http://www.informahealthcare.com/abb Artificial Cells, Blood Substitutes, and Biotechnology] Journal of ''Artificial Cells Blood Substitutes and Biotechnology'' | *[http://www.informahealthcare.com/abb Artificial Cells, Blood Substitutes, and Biotechnology] Journal of ''Artificial Cells Blood Substitutes and Biotechnology'' | ||
*Artificial Cells, Blood Substitutes, and Biotechnology Journal of Artificial Cells Blood Substitutes and Biotechnology | *Artificial Cells, Blood Substitutes, and Biotechnology Journal of Artificial Cells Blood Substitutes and Biotechnology | ||
| + | |||
== 编者推荐 == | == 编者推荐 == | ||
====[https://swarma.org/?p=13130 人类终极挑战:赋予人造细胞生命,全人工细胞实现临近]==== | ====[https://swarma.org/?p=13130 人类终极挑战:赋予人造细胞生命,全人工细胞实现临近]==== | ||
| + | 二十多年来,制造有生命特征的“人造细胞”是科学家执着的追求,而现在有生物学家预测:第一个全人工合成的细胞可能会在十年内出现。如果这个细胞还能成长、分裂,无疑那将是人类巨大的进步。 | ||
| + | |||
| + | |||
| + | ====[https://swarma.org/?p=25208 Cell重磅:十年磨一剑,科学家成功合成出可正常生长分裂的 “人造细胞”]==== | ||
| + | 人工细胞的合成对于科学家理解生命与基因的奥秘具有重要意义。20 多年以来,人们一直在尝试在实验室中,通过将一些生物分子拼凑起来从而创造出可以自主控制、代谢、维持生命的人造细胞。而现在,这一努力终于取得了重要突破。近日,来自美国 J. Craig Venter 研究所(JCVI),美国国家标准与技术研究院(NIST)以及麻省理工学院(MIT)的研究人员合作,成功创造出了一个非常简单的人工合成细胞,该细胞可以正常生长和分裂。 | ||
2022年3月20日 (日) 19:06的版本
人工细胞 artificial cell、人工合成细胞 synthetic cell或最小细胞 minimal cell是一种模仿生物细胞单个或多个功能的工程粒子。通常,人工细胞是包裹生物活性物质的生物膜或聚合物膜。[1] 因此,脂质体、高分子囊泡、纳米微粒、微胶囊和其他一些颗粒可以被定性为人工细胞。
”人工细胞”和“人工合成细胞”这两个术语用于各种不同的领域,可以具有不同的含义,它们也反映在文章的不同章节中。一些更严格的定义是基于这样的假设:“细胞”一词直接与生物细胞有关,因此这些结构必须是活的(或活的有机体的一部分) ,而“人工”一词则意味着这些结构是自下而上人工建造的,即源于基本组成部分。因此,在合成生物学领域,人工细胞可以被理解为一个完全合成的细胞,可以捕获能量,维持离子梯度,包含大分子,以及存储信息,并具有复制能力。[2]目前,这种人工细胞还没有被制造出来。
然而,在其他情况下,“人工”一词并不意味着整个结构是人造的,而是指一种生物细胞的某些功能或结构可以用合成实体取代或补充的想法。
在其他领域,“人工细胞”一词可指在尺寸或结构方面,一定程度上类似于生物细胞的,由非生物成分合成的,甚至是完全由非生物成分制成的任何隔室。术语“人工细胞”也用于具有直接应用的结构,例如用于药物传递的隔室。微囊化可以促进细胞膜内的新陈代谢,交换小分子,防止大型物质通过。[3][4] 包囊的主要优点包括改善体内的拟态,提高物质的溶解性和减少免疫反应。值得注意的是,人工细胞在临床上已成功应用于血液灌流。[5]
活人工细胞的底层工程
德国病理学家鲁道夫·韦尔乔夫 Rudolf Virchow提出了这样一个观点: 生命不仅来自细胞,而且每一个细胞都来自另一个细胞。[6]到目前为止,大多数创建人工细胞的尝试都只创建了能够模仿细胞特定任务的包。虽然无细胞转录和翻译反应的进展使许多基因得以表达,但这些努力与产生一个完全运作的细胞还有很远的距离。
自下而上构建人工细胞的方法涉及创建一个完全由非生物材料构成的原生质体。正如术语“细胞”所暗示的那样,一个前提是要生成某种定义了单个细胞单元的隔室。由于磷脂膜在所有活的生物细胞中起着选择性屏障的作用,[7] 因此可以选择磷脂膜作为一个明显的划分界限。科学家通过将生物分子包裹在细胞大小的磷脂小泡中,观察这些分子在生物细胞中的类似作用,从而重建某些细胞功能。[8]类似地,功能性生物构建模块可以被封装在这些脂质隔室中,以实现人工细胞的合成(无论多么初级)。
人们想到利用合成遗传信息,制备具有DNA自复制能力的磷脂双层囊泡。脂质膜的形成、通过模板过程进行的 DNA 和 RNA 复制、为了在脂膜上进行主动运输的化学能量的收集,是这种人工细胞的三个基本元素。[9][10]这个原始细胞将会遇到的主要障碍是创建一个最小的包含所有足够的生命信息的合成DNA,以及细胞发展中不可或缺的非遗传成分的复制,如分子自我组织。[11] 不过,人们希望通过这种自下而上的方法,更深入了解关于在细胞层面组织的基本问题和生物生命起源问题。迄今为止,还没有利用生命分子合成出能够自我繁殖的完全人工细胞,尽管目前各种团体正朝着这一目标努力,但它仍然在遥远的未来。[12]
另一种方法是,创造一个类似于人们认为在进化过程中存在的,称为原始汤的状态的原始细胞。各种RNA聚合物可以被包封在囊泡中,在如此小的边界条件下,将进行化学反应测试。[13]
伦理和争议
原始细胞的研究引起了争议和反对意见,包括对”人工生命”模糊定义的批评。[14]尽管对原细胞最普遍的担忧是它们通过不受控制的复制对人类健康和环境的潜在威胁,但创造一个基本的生命单位最紧迫的问题是伦理。[15]
国际研究
2010年代中期,研究界开始认识到有必要统一人工合成细胞的研究领域,承认从非生物成分构建一个完整生物体的任务超出了一个国家的资源范围。[16]
2017年,国际上大规模的人工合成活细胞建设研究合作正式启动,[17]随后若干国家也开展了国家间的合成细胞合作研究。这些全国性组织包括 FabriCell[18]、 MaxSynBio [19]和 BaSyC。[20]欧洲合成细胞的努力在2019年被统一为 SynCellEU 倡议。[21]
自上而下创建一个最小活细胞的方法
来自克莱格·凡特研究所 J. Craig Venter Institute的成员使用了一种自上而下的计算方法,将活生物体中的基因敲除至最小的一组基因。[22]2010年,研究小组利用人工合成中被认为是生命最低要求的 DNA,成功地创造了一个一株能复制的丝状支原体(支原体实验室) ,这种 DNA 被插入到一个基因组空白的细菌中。[22]人们希望,自上而下的生物合成过程将能够插入新的基因,这些基因将发挥有利的作用,例如产生氢气作为燃料或在大气中捕获过量的二氧化碳。[15]无数的调节、新陈代谢和信号网络没有被完全地表现,这些自上而下的方法对于理解基本的分子调控有局限性,因为宿主生物有一个复杂的和不完全确定的分子组成。[23]2019年,支原体 Syn3.0细胞中所有通路的完整计算模型被发表,这是第一个完整的微生物活体硅模型。[24]
大公司已经在生物领域投入了大量资金,比如与合成基因公司合作的埃克森美孚公司 ExxonMobil、克雷格·文特 Craig Venter的开发藻类燃料的生物合成公司[25]
截至2016年,生殖支原体 Mycoplasma genitalium是唯一一种可以作为最小细胞工程起点的有机体,因为它拥有已知最小的可以在实验室条件下培育的基因组; 野生型品种有482个基因,去除正好100个被认为非必需的基因,就能培育出生长速度提高的可存活菌株。减少基因组的大肠杆菌是更有用的,而且已经开发出去除了15% 的基因组的可生存菌株。[26]
通过将一个完全合成的基因组引入到基因组上空出的宿主细胞中,即创造出了一种人工细胞。[22]尽管由于宿主细胞的细胞质成分和细胞膜被保留下来而不完全是人工合成的,但该工程细胞处于合成基因组的控制之下,能够自复制。
医用人工细胞
历史
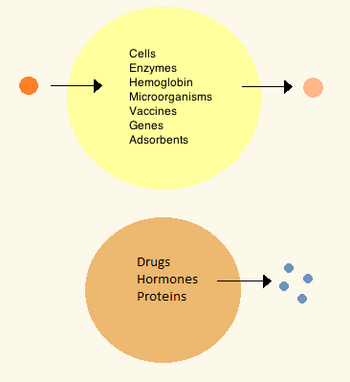
20世纪60年代,Thomas Chang研制出了微胶囊,由于它们是由人造材料制成的细胞大小的隔室,后来他称之为“人工细胞”。[27]这些细胞由尼龙、火棉胶或交联蛋白质的超薄膜组成,其半透性使得小分子可以扩散进出细胞。这些细胞是微米大小,含有细胞,酶,血红蛋白,磁性材料,吸附剂和蛋白质。[3]
后来人工细胞的尺寸从百微米到纳米不等,可以携带微生物、疫苗、基因、药物、激素和多肽。[3] 人工细胞的第一次临床应用是以药用活性炭为包囊进行血液灌流。[28]
在20世纪70年代,研究人员能够将酶、蛋白质和激素引入到可生物降解的微胶囊中,后来这种微胶囊在诸如莱希-尼亨氏症候群 Lesch–Nyhan syndrome之类的疾病中得到临床应用。[29]尽管Thomas Chang最初的研究集中在人工红细胞上,但直到20世纪90年代中期,才出现了可生物降解的人工红细胞。[30]1994年,生物细胞封装的人工细胞首次在临床上用于治疗糖尿病患者,[31]此后,其他类型的细胞,如肝细胞、成体干细胞和基因工程细胞已被封装,并正在研究用于组织再生。[32][33]
材料
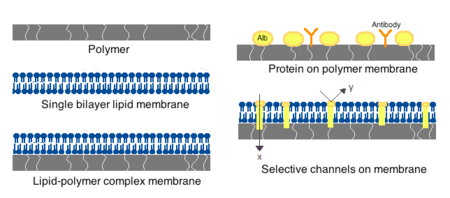
用于人工细胞的膜可以由简单的聚合物、交联蛋白质、脂质膜或聚合物-脂质复合物制成。此外,膜可以被设计成表面蛋白如白蛋白、抗原、 Na/K-ATP 酶载体或孔隙如离子通道。常用的水凝胶聚合物如海藻酸钠、纤维素和热塑性聚合物如甲基丙烯酸羟乙酯-甲基丙烯酸甲酯(HEMA-MMA)、聚丙烯腈-聚氯乙烯(PAN-PVC) ,以及上述各种聚合物的变异。[4]所用的材料决定了细胞膜的渗透性,对于聚合物来说,这取决于截留分子量(MWCO)。[4]MWCO 是可以自由通过孔隙的分子的最大分子量,对于确定营养物质、废物和其他临界分子的充分扩散很重要。亲水性聚合物具有生物相容性的潜力,可以制成多种形式,包括聚合物胶束、溶胶-凝胶混合物、物理共混物以及交联粒子和纳米粒子。[4]特别令人感兴趣的是对pH值或温度变化有反应的刺激性聚合物,用于靶向传递。由于 pH 值和温度的不同,这些聚合物可以通过宏观注射和凝固或原位凝胶以液体形式给药。纳米颗粒和脂质体制剂也常用于材料的包封和递送。脂质体的一个主要优点是它们能够与细胞膜和细胞器膜融合。
制备
人工细胞的制备和包封已经发展出许多不同的方法。通常,如纳米粒子、聚合物或脂质体等小泡被合成。乳化液通常是通过高压设备,如高压匀浆器或微射流机来制造的。下面将介绍硝化纤维素的两种微胶囊化方法.
高压均质化 High-pressure homogenization
在高压匀浆器中,油/液悬浮液中的两种液体在很高的压力下通过一个小孔。这一过程划分了产品,并允许产生极细的颗粒,小到1nm。
微射流处理 Microfluidization
微射流技术使用专利的微射流机获得更多的均质悬浮液,可以产生比匀浆器更小的颗粒。匀浆器首先被用来制造粗颗粒悬浮液,然后在高压下被泵入微射流机。然后将流体分成两股流体,这两股流体将在相互作用室中以非常高的速度反应,直到得到所需的粒子尺寸。[34]这种技术允许大规模生产磷脂脂质体和后续材料纳米包埋。
点滴法 Drop method
在这种方法中,细胞溶液滴入硝酸纤维素的火棉胶溶液中。当液滴通过火棉胶时,由于火棉胶的界面聚合特性,它被涂上了一层膜。细胞随后沉积在石蜡中,细胞膜凝固,最后悬浮在盐溶液中。点滴法用于制造大型人工细胞,包裹生物细胞、干细胞和基因工程干细胞。
乳化法 Emulsion method
乳化法的不同之处在于,要被包裹的材料通常较小,被放置在反应室的底部,在反应室的顶部加入火棉胶并离心,或以其他方式搅拌以便形成乳化液。然后将胶囊材料分散并悬浮在盐溶液中。
材料
药物的释放和运送
用于药物运送的人工细胞与其他人工细胞不同,是由于其内容物要扩散出细胞膜,或者被宿主靶细胞吞噬和消化。通常使用的是亚微米,脂质膜人工细胞,可称为纳米胶囊,纳米粒子,聚合体,或其他变体。
酶疗法 Enzyme therapy
目前正在积极研究酶疗法,用于治疗有某种酶过度表达、表达不足、有缺陷或根本不存在等表现的遗传代谢性疾病。在有缺陷的酶的表达不足或表达不足的情况下,活性形式的该种酶将被引入人体以弥补缺陷。另一方面,酶的过度表达可以通过引入竞争性的非功能性酶来抵消,也就是说,一种将底物代谢为非活性产物的酶。当置于人工细胞中时,酶可以比游离酶更长时间地发挥作用,[3]并且可以通过聚合物接合进一步优化。[35]
人工细胞包裹法研究的第一种酶是治疗小鼠淋巴肉瘤的天冬酰胺酶。[36]这种治疗延缓了肿瘤的发生和生长。[37]这些初步的发现导致了在酪氨酸依赖性黑色素瘤中使用人工细胞进行酶传递的进一步研究。这些肿瘤的生长比正常细胞更依赖于酪氨酸,研究表明,降低小鼠全身酪氨酸水平可以抑制黑色素瘤的生长。[38]利用人工细胞输送酪氨酸酶和消化酪氨酸的酶,可以提高酶的稳定性,并且能有效地去除酪氨酸,而不会产生与饮食中酪氨酸恶化有关的严重副作用。[39]
在某些癌症中,人工细胞酶疗法对于激活诸如异环磷酰胺之类的前药也很有意义。将细胞色素 p450酶包裹在人工细胞中,将其转化为活性药物,可以特制地在胰腺癌中积累,或将人工细胞移植到肿瘤部位附近。在这里,被激活的异环磷酰胺的局部浓度将远远高于身体其他部位,从而防止全身中毒。[40]这种治疗在动物身上取得了成功,[41]在 I/II 期临床试验中,晚期胰腺癌患者的生存率中位数增加了一倍,一年生存率增加了两倍。[40]
基因疗法 Gene therapy
在基因疾病的治疗中,基因疗法旨在插入、改变或移除受影响个体细胞内的基因。该技术严重依赖于病毒载体,这引起了对插入突变和系统免疫反应的关注,它们在临床试验中导致了人类死亡[42][43]和白血病的发展[44][45]。利用裸DNA或质粒DNA作为载体载体本身的传递系统,避免了载体的需要,同时也遇到了系统传递效率低、组织靶向性差等问题。[4]
人工细胞被认为是一种非病毒载体,将转基因非自体细胞包裹起来并植入体内以表达重组蛋白。[46]通过传递含有小鼠生长激素的人工细胞,挽救了突变小鼠的生长迟缓,这种免疫分离已经被证明是有效的。[47]一些治疗胰腺癌、脊髓侧索硬化症和疼痛控制的方案已经进入人体临床试验阶段。[4]
血液灌流 Hemoperfusion
临床上第一次使用人工细胞的方法是用活性炭包裹血液灌流。[28]药用活性炭具有吸附许多大分子的能力,长期以来因其在意外中毒或过量服用中能够去除血液中的有毒物质而闻名。然而,通过直接使用木炭进行灌注是有毒的,因为它会导致血栓和血细胞损伤,随后被血小板清除。[48]人工细胞允许毒素扩散到细胞中,同时将危险物质保存在超薄的膜内。[28]
与血液透析相比,人工细胞血液灌流被认为是一种成本更低、效率更高的解毒方法。[3]在血液灌流中,成千上万的吸附性人工细胞通过在病人血液灌流的两端使用两个筛子被保存在一个小容器内。随着血液循环,毒素或药物扩散进入细胞,并被吸收材料所保留。人工细胞的细胞膜比透析用细胞的细胞膜要薄得多,细胞膜的小体积意味着细胞膜的表面积很大。这意味着一部分细胞理论上的质量传递比整个人工肾脏机器的质量传递高百倍。[3]该装置已被确立为治疗意外或自杀性中毒患者的常规临床方法,但也被引入治疗肝功能衰竭和肾功能衰竭,以实现这些器官的部分功能。[3]人工细胞血液灌流也被提议用于免疫吸附,通过在人工细胞表面粘附免疫吸附材料,如白蛋白,可将抗体从体内除去。这一原理已被应用于从骨髓移植患者血浆中去除血型抗体,[49]以及通过单克隆抗体去除低密度脂蛋白治疗高胆固醇血症。[50]血液灌流在血液透析制造业薄弱的国家尤其有用,因为那里的血液透析设备往往更便宜,而且用于肾衰竭患者。
微囊细胞
最常用的制备人工细胞的方法是细胞包裹。微囊细胞通常由液体细胞悬浮液中产生控制尺寸的液滴获取,然后快速凝固或凝胶化以提供额外的稳定性。稳定化可以通过温度的变化或材料的交联来实现。细胞的微环境在包裹后发生变化,且通常从单分子层到悬浮在聚合物膜内的聚合物支架上。这种技术的缺点是微囊细胞会降低其生存能力以及增殖和分化能力。[51]此外,在微囊中待一段时间后,细胞会形成团簇,抑制氧气和代谢废物的交换,[52]导致细胞凋亡和坏死,从而限制细胞的功效,激活宿主的免疫系统。人工细胞已经成功地移植了一些细胞,包括治疗糖尿病的胰岛、[53]甲状旁腺细胞和肾上腺皮质细胞。
微囊肝细胞 Encapsulated hepatocytes
器官捐献者的短缺使人工细胞成为治疗肝衰竭替代疗法的关键。人工细胞用于肝细胞移植已被证实在动物肝病模型和生物人工肝装置中提供肝功能的可行性和有效性。[54]实验中,肝细胞附着在微载体表面,[55]然后进化成包裹在由聚赖氨酸外皮覆盖的海藻酸钠微滴中的三维基质中的肝细胞。这种给药方法的一个关键优势是在治疗期间避免了免疫抑制治疗。微囊肝细胞已被提议用于生物人工肝。该装置由一个嵌有分离肝细胞的圆柱形腔室组成,病人的血浆通过这个腔室在一种血液灌流中循环。由于微囊具有很高的比表面积和体积比,它们为基质扩散提供了较大的表面积,并且可以容纳大量的肝细胞。对诱导性肝衰竭小鼠的治疗显示存活率显著提高。[54]人工肝系统仍处于早期发展阶段,但对于等待器官移植或者病人自身的肝脏重新充分恢复正常功能的病人来说,显示出了潜力。迄今为止,应用人工肝系统和肝细胞移植治疗终末期肝病的临床试验表明,健康指标有所改善,但还没有提高存活率。[56]移植后人工肝细胞的短寿命和聚集是目前肝移植面临的主要障碍。[57]与干细胞一同包裹的肝细胞在培养和植入后显示出更高的存活率,单独植入人工干细胞也可以促成肝的再生。[58]因此,在再生医学中利用干细胞封装的研究引起了人们的兴趣。
微囊细菌细胞 Encapsulated bacterial cells
口服活菌群,目前正用于治疗调节肠道菌群,[59]预防腹泻疾病,[60]治疗幽门螺杆菌感染,特应性炎症,[61]乳糖不耐受[62]和免疫调节[63]等症病。虽然其作用机制尚未得到充分理解,但被认为其具有两个主要作用。首先是营养效应,即细菌与产毒细菌竞争。第二是卫生效应,即刺激抵抗定殖和免疫反应。[4]由于细菌培养物往往是免疫系统的目标,通常会在口服时被破坏,口服给药的方式便成了困难。人工细胞通过向体内提供拟态,以及其可选择的或长期的释放,来提高进入胃肠道系统的细菌的生存能力,从而帮助解决这些问题。[4]此外,还可以设计活细菌包囊,使小分子(包括多肽)扩散到体内,以用于治疗。[4]醋酸纤维素和海藻酸盐的变体等已被证明,可以成功用于细菌传递的膜。[4]微囊细菌细胞的其他用途还包括保护细菌细胞免受结核分枝杆菌的攻击,[64]以及促进免疫系统产生Ig分泌细胞。[65]该技术受到系统性感染、不良代谢活动和基因转移风险的限制。[4]然而,更大的挑战仍然是如何将足够多的有生命的细菌运送到目标部位。[4]
人工血细胞作为氧载体 Artificial blood cells as oxygen carriers
人工血细胞作为氧载体被用作一种红细胞替代品,尽管它们缺乏红细胞的其他成分。它们由一种合成的聚合体或围绕着纯化的动物、人或重组的血红蛋白的人工膜组成。[66]总的来说,血红蛋白的输送仍然是一个挑战,因为它在没有任何修饰的情况下输送时是剧毒的。在一些临床试验中,人们观察到血管升压效应。[67][68]
人工红细胞 Artificial red blood cells
在1980年代的艾滋病恐慌之后,人们对使用人工红细胞制造血液产生了研究兴趣。除了避开疾病传播的可能性之外,人工红细胞也是人们所需要的,因为它们可以消除与同种异体输血相关的缺点,如血型鉴定、免疫反应及其短暂的42天储存期。血红蛋白替代物可以存放在室温下,而不是在冷藏条件下存放一年以上。[3]人们已经尝试开发出一种完整工作的红细胞,它不仅包含碳酸,而且包含氧载体和与细胞相关的酶。第一次尝试是在1957年,用超薄聚合物膜[69]代替红细胞膜,然后通过脂质膜[70]和最近的可生物降解聚合物膜进行包封。[3]包括脂类和相关蛋白在内的生物红细胞膜还可以通过绕过巨噬细胞摄取和系统清除来包裹纳米颗粒和增加体内滞留时间。[71]
人工白细胞聚合体 Artificial leuko-polymersomes
人工白细胞聚合体是一种具有白细胞粘附特性的高分子囊泡。[72]高分子囊泡是由双层薄片组成的囊泡,可以包裹许多活性分子,如药物或酶。通过将白细胞的粘附特性添加到细胞膜上,白细胞可以减慢速度,或者在快速流动的循环系统内沿着上皮细胞壁滚动。
非常规类型的人工细胞
电子人工细胞 Electronic artificial cell
电子人工细胞的概念在2004年至2015年由约翰 · 麦卡斯基尔协调的一系列欧盟项目中得到扩展。
2004年至2008年,欧洲联盟委员会赞助制定了可编程人工细胞进化方案[73],其目标是为最终融入信息系统奠定基础,创建”由简单的有机和无机物质构成的微观自组织、自我复制和可进化的自主实体,这些物质可通过遗传程序进行编程,以履行特定功能”[73]。PACE 项目开发了第一台欧米茄机器,这是一种人工细胞的微流体生命支持系统,可以补充化学缺失的功能(最初由Norman Packard、Steen Rasmussen、Mark Beadau和John McCaskill基尔提出)。最终目标是在复杂的微型可编程环境中实现可进化的混合细胞。欧米茄机器的功能可以逐步被移除,这对人工细胞化学提出了一系列可解决的进化挑战。该项目实现了人工细胞三个核心功能(遗传子系统、包容系统和新陈代谢系统)成对的化学整合,并为包容和基因扩增的整合创造了新的空间分辨可编程微流体环境。[73]这个项目导致了欧洲生物技术中心的建立。[74]
在这项研究之后,2007年,John McCaskill基尔提议集中研究一种电子补充型人工电池,称为电子化学电池。关键的想法是在一个二维薄膜中,使用一个大规模并行处理机阵列的电极与局部专用电子电路耦合,以补充新出现的化学细胞功能。定义电极切换和传感电路的局部电子信息可以作为电子基因组,补充新协议中的分子序列信息。与欧洲联盟委员会的一项研究提案取得了成功,与计算机设备行动伙伴关系联盟部分重叠的一个国际科学家小组开始了2008-2012年电子化学细胞项目的工作。除其他外,该项目表明,电子控制的特定序列的局部转移可以用作未来人工细胞遗传增殖的人工空间控制系统,并且新陈代谢的核心过程可以通过适当的涂层电极阵列传递。
这种方法的主要局限性,除了最初在掌握微观电化学和电动力学方面的困难之外,是把电子系统作为一种刚性的、非自主的宏观硬件连接在一起。2011年,麦卡斯基尔提议颠倒电子学和化学的几何学: 不把化学物质放在活跃的电子介质中,而是把微观的自主电子学放在化学介质中。他组织了一个项目,以解决第三代100微米规模的电子人工细胞问题,这种细胞可以由两个半细胞”实验室”自我组装,以封闭内部化学空间,并借助浸入其中的有源电子设备发挥功能。这些细胞可以复制它们的电子和化学成分,并且能够在它们特殊的预合成微观构件所提供的约束条件下进化。2012年9月,这个项目开始了工作。[75]
杰瓦努 Jeewanu
杰瓦努原始细胞是具有细胞样结构的合成化学粒子,似乎具有一定的功能活性。[76]1963年首次在阳光下由简单矿物质和基本有机物合成,据报道它仍然具有一些新陈代谢能力,包括半透膜、氨基酸、磷脂、碳水化合物和类 RNA分子。然而,杰瓦努的性质和属性仍有待澄清。[76][77] However, the nature and properties of the Jeewanu remains to be clarified.[76][77][78]
See also
参考文献
- ↑ Buddingh' BC, van Hest JC (April 2017). "Artificial Cells: Synthetic Compartments with Life-like Functionality and Adaptivity". Accounts of Chemical Research. 50 (4): 769–777. doi:10.1021/acs.accounts.6b00512. PMC 5397886. PMID 28094501.
- ↑ Deamer D (July 2005). "A giant step towards artificial life?". Trends in Biotechnology. 23 (7): 336–338. doi:10.1016/j.tibtech.2005.05.008. PMID 15935500.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Artificial cells : biotechnology, nanomedicine, regenerative medicine, blood substitutes, bioencapsulation, cell/stem cell therapy. Hackensack, N.J.: World Scientific. 2007. ISBN 978-981-270-576-1.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 Artificial cells, cell engineering and therapy.. Boca Raton, Fl: Woodhead Publishing Limited. 2007. ISBN 978-1-84569-036-6.
- ↑ Polymeric materials and artificial organs based on a symposium sponsored by the Division of Organic Coatings and Plastics Chemistry at the 185th Meeting of the American Chemical Society. Washington, D.C.: American Chemical Society. 1983. ISBN 978-0-8412-1084-4.
- ↑ (in de) Die cellularpathologie in ihrer begründung auf physiologische und pathologische gewebelehre. Zwanzig Vorlesungen gehalten wahrend der Monate Februar, Marz und April 1858. Berlin: Verlag von August Hirschwald. 1858. p. xv. https://archive.org/details/diecellularpatho1858virc.
- ↑ Kamiya K, Takeuchi S (August 2017). "Giant liposome formation toward the synthesis of well-defined artificial cells". Journal of Materials Chemistry B. 5 (30): 5911–5923. doi:10.1039/C7TB01322A. PMID 32264347.
- ↑ Litschel T, Schwille P (May 2021). "Protein Reconstitution Inside Giant Unilamellar Vesicles". Annual Review of Biophysics. 50: 525–548. doi:10.1146/annurev-biophys-100620-114132. PMID 33667121. S2CID 232131463.
- ↑ Szostak JW, Bartel DP, Luisi PL (January 2001). "Synthesizing life". Nature. 409 (6818): 387–390. doi:10.1038/35053176. PMID 11201752. S2CID 4429162.
- ↑ Pohorille A, Deamer D (March 2002). "Artificial cells: prospects for biotechnology". Trends in Biotechnology. 20 (3): 123–128. doi:10.1016/S0167-7799(02)01909-1. hdl:2060/20020043286. PMID 11841864.
- ↑ Noireaux V, Maeda YT, Libchaber A (March 2011). "Development of an artificial cell, from self-organization to computation and self-reproduction". Proceedings of the National Academy of Sciences of the United States of America. 108 (9): 3473–3480. Bibcode:2011PNAS..108.3473N. doi:10.1073/pnas.1017075108. PMC 3048108. PMID 21317359.
- ↑ Rasmussen S, Chen L, Nilsson M, Abe S (Summer 2003). "Bridging nonliving and living matter". Artificial Life. 9 (3): 269–316. CiteSeerX 10.1.1.101.1606. doi:10.1162/106454603322392479. PMID 14556688. S2CID 6076707.
- ↑ Gilbert W (20 February 1986). "Origin of life: The RNA world". Nature. 319 (6055): 618. Bibcode:1986Natur.319..618G. doi:10.1038/319618a0. S2CID 8026658.
- ↑ Bedau M, Church G, Rasmussen S, Caplan A, Benner S, Fussenegger M, et al. (May 2010). "Life after the synthetic cell". Nature. 465 (7297): 422–424. Bibcode:2010Natur.465..422.. doi:10.1038/465422a. PMID 20495545. S2CID 27471255.
- ↑ 15.0 15.1 The ethics of protocells moral and social implications of creating life in the laboratory ([Online-Ausg.] ed.). Cambridge, Mass.: MIT Press. 2009. ISBN 978-0-262-51269-5.
- ↑ "From chemicals to life: Scientists try to build cells from scratch". Retrieved 4 Dec 2019.
- ↑ "Build-a-Cell". Retrieved 4 Dec 2019.
- ↑ "FabriCell". Retrieved 8 Dec 2019.
- ↑ "MaxSynBio - Max Planck Research Network in Synthetic Biology". Retrieved 8 Dec 2019.
- ↑ "BaSyC". Retrieved 8 Dec 2019.
- ↑ "SynCell EU". Retrieved 8 Dec 2019.
- ↑ 22.0 22.1 22.2 Gibson DG, Glass JI, Lartigue C, Noskov VN, Chuang RY, Algire MA, et al. (July 2010). "Creation of a bacterial cell controlled by a chemically synthesized genome". Science. 329 (5987): 52–56. Bibcode:2010Sci...329...52G. doi:10.1126/science.1190719. PMID 20488990. S2CID 7320517.
- ↑ Armstrong R (September 2014). "Designing with protocells: applications of a novel technical platform". Life. 4 (3): 457–490. doi:10.3390/life4030457. PMC 4206855. PMID 25370381.
- ↑ Breuer M, Earnest TM, Merryman C, Wise KS, Sun L, Lynott MR, et al. (January 2019). "Essential metabolism for a minimal cell". eLife. 8. doi:10.7554/eLife.36842. PMC 6609329. PMID 30657448.
- ↑ Sheridan C (September 2009). "Big oil bucks for algae". Nature Biotechnology. 27 (9): 783. doi:10.1038/nbt0909-783. PMID 19741613. S2CID 205270805.
- ↑ EU Directorate-General for Health and Consumers (2016-02-12). "Opinion on synthetic biology II: Risk assessment methodologies and safety aspects" (in English). Publications Office. doi:10.2772/63529.
{{cite journal}}: Cite journal requires|journal=(help) - ↑ Chang TM (October 1964). "SEMIPERMEABLE MICROCAPSULES". Science. 146 (3643): 524–525. Bibcode:1964Sci...146..524C. doi:10.1126/science.146.3643.524. PMID 14190240. S2CID 40740134.
- ↑ 28.0 28.1 28.2 Chang TM (1996). "Editorial: past, present and future perspectives on the 40th anniversary of hemoglobin based red blood cell substitutes". Artificial Cells Blood Substit Immobil Biotechnol. 24: ixxxvi.
- ↑ Palmour RM, Goodyer P, Reade T, Chang TM (September 1989). "Microencapsulated xanthine oxidase as experimental therapy in Lesch-Nyhan disease". Lancet. 2 (8664): 687–688. doi:10.1016/s0140-6736(89)90939-2. PMID 2570944. S2CID 39716068.
- ↑ Blood substitutes. Basel: Karger. 1997. ISBN 978-3-8055-6584-4.
- ↑ Soon-Shiong P, Heintz RE, Merideth N, Yao QX, Yao Z, Zheng T, et al. (April 1994). "Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation". Lancet. 343 (8903): 950–951. doi:10.1016/S0140-6736(94)90067-1. PMID 7909011. S2CID 940319.
- ↑ Liu ZC, Chang TM (June 2003). "Coencapsulation of hepatocytes and bone marrow stem cells: in vitro conversion of ammonia and in vivo lowering of bilirubin in hyperbilirubemia Gunn rats". The International Journal of Artificial Organs. 26 (6): 491–497. doi:10.1177/039139880302600607. PMID 12894754. S2CID 12447199.
- ↑ Aebischer P, Schluep M, Déglon N, Joseph JM, Hirt L, Heyd B, et al. (June 1996). "Intrathecal delivery of CNTF using encapsulated genetically modified xenogeneic cells in amyotrophic lateral sclerosis patients". Nature Medicine. 2 (6): 696–699. doi:10.1038/nm0696-696. PMID 8640564. S2CID 8049662.
- ↑ Vivier A, Vuillemard JC, Ackermann HW, Poncelet D (1992). "Large-scale blood substitute production using a microfluidizer". Biomaterials, Artificial Cells, and Immobilization Biotechnology. 20 (2–4): 377–397. doi:10.3109/10731199209119658. PMID 1391454.
- ↑ Park et al. 1981
- ↑ Chang TM (January 1971). "The in vivo effects of semipermeable microcapsules containing L-asparaginase on 6C3HED lymphosarcoma". Nature. 229 (5280): 117–118. Bibcode:1971Natur.229..117C. doi:10.1038/229117a0. PMID 4923094. S2CID 4261902.
- ↑ Yu B, Chang TM (April 2004). "Effects of long-term oral administration of polymeric microcapsules containing tyrosinase on maintaining decreased systemic tyrosine levels in rats". Journal of Pharmaceutical Sciences. 93 (4): 831–837. doi:10.1002/jps.10593. PMID 14999721.
- ↑ Meadows GG, Pierson HF, Abdallah RM, Desai PR (August 1982). "Dietary influence of tyrosine and phenylalanine on the response of B16 melanoma to carbidopa-levodopa methyl ester chemotherapy". Cancer Research. 42 (8): 3056–3063. PMID 7093952.
- ↑ Chang TM (February 2004). "Artificial cell bioencapsulation in macro, micro, nano, and molecular dimensions: keynote lecture". Artificial Cells, Blood Substitutes, and Immobilization Biotechnology. 32 (1): 1–23. doi:10.1081/bio-120028665. PMID 15027798. S2CID 37799530.
- ↑ 40.0 40.1 Löhr M, Hummel F, Faulmann G, Ringel J, Saller R, Hain J, et al. (May 2002). "Microencapsulated, CYP2B1-transfected cells activating ifosfamide at the site of the tumor: the magic bullets of the 21st century". Cancer Chemotherapy and Pharmacology. 49 (Suppl 1): S21–S24. doi:10.1007/s00280-002-0448-0. PMID 12042985. S2CID 10329480.
- ↑ Kröger JC, Benz S, Hoffmeyer A, Bago Z, Bergmeister H, Günzburg WH, et al. (1999). "Intra-arterial instillation of microencapsulated, Ifosfamide-activating cells in the pig pancreas for chemotherapeutic targeting". Pancreatology. 3 (1): 55–63. doi:10.1159/000069147. PMID 12649565. S2CID 23711385.
- ↑ Carmen IH (April 2001). "A death in the laboratory: the politics of the Gelsinger aftermath". Molecular Therapy. 3 (4): 425–428. doi:10.1006/mthe.2001.0305. PMID 11319902.
- ↑ Raper SE, Chirmule N, Lee FS, Wivel NA, Bagg A, Gao GP, et al. (1 September 2003). "Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer". Molecular Genetics and Metabolism. 80 (1–2): 148–158. doi:10.1016/j.ymgme.2003.08.016. PMID 14567964.
- ↑ Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P, et al. (April 2000). "Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease". Science. 288 (5466): 669–672. Bibcode:2000Sci...288..669C. doi:10.1126/science.288.5466.669. PMID 10784449.
- ↑ Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack MP, Wulffraat N, Leboulch P, et al. (October 2003). "LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1". Science. 302 (5644): 415–419. Bibcode:2003Sci...302..415H. doi:10.1126/science.1088547. PMID 14564000. S2CID 9100335.
- ↑ Chang PL, Van Raamsdonk JM, Hortelano G, Barsoum SC, MacDonald NC, Stockley TL (February 1999). "The in vivo delivery of heterologous proteins by microencapsulated recombinant cells". Trends in Biotechnology. 17 (2): 78–83. doi:10.1016/S0167-7799(98)01250-5. PMID 10087608.
- ↑ al-Hendy A, Hortelano G, Tannenbaum GS, Chang PL (February 1995). "Correction of the growth defect in dwarf mice with nonautologous microencapsulated myoblasts--an alternate approach to somatic gene therapy". Human Gene Therapy. 6 (2): 165–175. doi:10.1089/hum.1995.6.2-165. PMID 7734517.
- ↑ Dunea G, Kolff WJ (1965). "CLINICAL EXPERIENCE WITH THE YATZIDIS CHARCOAL ARTIFICIAL KIDNEY". Transactions - American Society for Artificial Internal Organs. 11: 178–182. doi:10.1097/00002480-196504000-00035. PMID 14329080.
- ↑ Bensinger WI, Buckner CD, Clift RA (1985). "Whole blood immunoadsorption of anti-A or anti-B antibodies". Vox Sanguinis. 48 (6): 357–361. doi:10.1111/j.1423-0410.1985.tb00196.x. PMID 3892895. S2CID 12777645.
- ↑ Yang L, Cheng Y, Yan WR, Yu YT (2004). "Extracorporeal whole blood immunoadsorption of autoimmune myasthenia gravis by cellulose tryptophan adsorbent". Artificial Cells, Blood Substitutes, and Immobilization Biotechnology. 32 (4): 519–528. doi:10.1081/bio-200039610. PMID 15974179. S2CID 7269229.
- ↑ "Calcium phosphate-mediated DNA transfection". Gene Therapeutics. Boston: Birkhauser. 1994. pp. 157–179. doi:10.1007/978-1-4684-6822-9_9. ISBN 978-1-4684-6822-9.
- ↑ Ponce S, Orive G, Gascón AR, Hernández RM, Pedraz JL (April 2005). "Microcapsules prepared with different biomaterials to immobilize GDNF secreting 3T3 fibroblasts". International Journal of Pharmaceutics. 293 (1–2): 1–10. doi:10.1016/j.ijpharm.2004.10.028. PMID 15778039.
- ↑ Kizilel S, Garfinkel M, Opara E (December 2005). "The bioartificial pancreas: progress and challenges". Diabetes Technology & Therapeutics. 7 (6): 968–985. doi:10.1089/dia.2005.7.968. PMID 16386103.
- ↑ 54.0 54.1 Dixit V, Gitnick G (27 November 2003). "The bioartificial liver: state-of-the-art". The European Journal of Surgery. Supplement. 164 (582): 71–76. doi:10.1080/11024159850191481. PMID 10029369.
- ↑ Demetriou AA, Whiting JF, Feldman D, Levenson SM, Chowdhury NR, Moscioni AD, et al. (September 1986). "Replacement of liver function in rats by transplantation of microcarrier-attached hepatocytes". Science. 233 (4769): 1190–1192. Bibcode:1986Sci...233.1190D. doi:10.1126/science.2426782. PMID 2426782.
- ↑ Sgroi A, Serre-Beinier V, Morel P, Bühler L (February 2009). "What clinical alternatives to whole liver transplantation? Current status of artificial devices and hepatocyte transplantation". Transplantation. 87 (4): 457–466. doi:10.1097/TP.0b013e3181963ad3. PMID 19307780.
- ↑ Liu ZC, Chang TM (March 2002). "Increased viability of transplanted hepatocytes when hepatocytes are co-encapsulated with bone marrow stem cells using a novel method". Artificial Cells, Blood Substitutes, and Immobilization Biotechnology. 30 (2): 99–112. doi:10.1081/bio-120003191. PMID 12027231. S2CID 26667880.
- ↑ Therapeutic applications of cell microencapsulation (Online-Ausg. ed.). New York: Springer Science+Business Media. 2010. ISBN 978-1-4419-5785-6.
- ↑ Mattila-Sandholm T, Blum S, Collins JK, Crittenden R, De Vos W, Dunne C, et al. (1 December 1999). "Probiotics: towards demonstrating efficacy". Trends in Food Science & Technology. 10 (12): 393–399. doi:10.1016/S0924-2244(00)00029-7.
- ↑ Huang JS, Bousvaros A, Lee JW, Diaz A, Davidson EJ (November 2002). "Efficacy of probiotic use in acute diarrhea in children: a meta-analysis". Digestive Diseases and Sciences. 47 (11): 2625–2634. doi:10.1023/A:1020501202369. PMID 12452406. S2CID 207559325.
- ↑ Isolauri E, Arvola T, Sütas Y, Moilanen E, Salminen S (November 2000). "Probiotics in the management of atopic eczema". Clinical and Experimental Allergy. 30 (11): 1604–1610. doi:10.1046/j.1365-2222.2000.00943.x. PMID 11069570. S2CID 13524021.
- ↑ Lin MY, Yen CL, Chen SH (January 1998). "Management of lactose maldigestion by consuming milk containing lactobacilli". Digestive Diseases and Sciences. 43 (1): 133–137. doi:10.1023/A:1018840507952. PMID 9508514. S2CID 22890925.
- ↑ Gill HS (1 May 1998). "Stimulation of the Immune System by Lactic Cultures". International Dairy Journal. 8 (5–6): 535–544. doi:10.1016/S0958-6946(98)00074-0.
- ↑ Aldwell FE, Tucker IG, de Lisle GW, Buddle BM (January 2003). "Oral delivery of Mycobacterium bovis BCG in a lipid formulation induces resistance to pulmonary tuberculosis in mice". Infection and Immunity. 71 (1): 101–108. doi:10.1128/IAI.71.1.101-108.2003. PMC 143408. PMID 12496154.
- ↑ Park JH, Um JI, Lee BJ, Goh JS, Park SY, Kim WS, Kim PH (September 2002). "Encapsulated Bifidobacterium bifidum potentiates intestinal IgA production". Cellular Immunology. 219 (1): 22–27. doi:10.1016/S0008-8749(02)00579-8. PMID 12473264.
- ↑ Kim HW, Greenburg AG (September 2004). "Artificial oxygen carriers as red blood cell substitutes: a selected review and current status". Artificial Organs. 28 (9): 813–828. doi:10.1111/j.1525-1594.2004.07345.x. PMID 15320945.
- ↑ "Blood and HemAssistTM (DCLHb): Potentially a complementary therapeutic team". Blood Substitutes: Principles, Methods, Products and Clinical Trials. 2. Basel: Karger. 1998. pp. 39–57.
- ↑ Burhop KE, Estep TE (2001). "Hemoglobin induced myocardial lesions". Artificial Cells, Blood Substitutes, and Biotechnology. 29 (2): 101–106. doi:10.1080/10731190108951271. PMC 3555357.
- ↑ "30th Anniversary in Artificial Red Blood Cell Research". Artificial Cells, Blood Substitutes and Biotechnology. 16 (1–3): 1–9. 1 January 1988. doi:10.3109/10731198809132551.
- ↑ Djordjevich L, Miller IF (May 1980). "Synthetic erythrocytes from lipid encapsulated hemoglobin". Experimental Hematology. 8 (5): 584–592. PMID 7461058.
- ↑ Hu CM, Zhang L, Aryal S, Cheung C, Fang RH, Zhang L (July 2011). "Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform". Proceedings of the National Academy of Sciences of the United States of America. 108 (27): 10980–10985. Bibcode:2011PNAS..10810980H. doi:10.1073/pnas.1106634108. PMC 3131364. PMID 21690347.
- ↑ Hammer DA, Robbins GP, Haun JB, Lin JJ, Qi W, Smith LA, et al. (1 January 2008). "Leuko-polymersomes". Faraday Discussions. 139: 129–41, discussion 213–28, 419–20. Bibcode:2008FaDi..139..129H. doi:10.1039/B717821B. PMC 2714229. PMID 19048993.
- ↑ 73.0 73.1 73.2 "Programmable Artificial Cell Evolution" (PACE)". PACE Consortium.
- ↑ "European center for living technology". European Center for Living Technology. Archived from the original on 2011-12-14.
- ↑ "Microscale Chemically Reactive Electronic Agents". Ruhr Universität Bochum.
- ↑ 76.0 76.1 76.2 Grote M (September 2011). "Jeewanu, or the 'particles of life'. The approach of Krishna Bahadur in 20th century origin of life research" (PDF). Journal of Biosciences. 36 (4): 563–570. doi:10.1007/s12038-011-9087-0. PMID 21857103. S2CID 19551399. Archived from the original (PDF) on 2014-03-23.
- ↑ 77.0 77.1 Gupta VK, Rai RK (2013). "Histochemical localisation of RNA-like material in photochemically formed self-sustaining, abiogenic supramolecular assemblies 'Jeewanu'". Int. Res. J. Of Science & Engineering. 1 (1): 1–4. ISSN 2322-0015.
- ↑ Caren, Linda D.; Ponnamperuma, Cyril (1967). "A review of some experiments on the synthesis of 'Jeewanu'" (PDF). NASA Technical Memorandum X-1439.
外部链接
- Artificial Cells, Blood Substitutes, and Biotechnology Journal of Artificial Cells Blood Substitutes and Biotechnology
- Artificial Cells, Blood Substitutes, and Biotechnology Journal of Artificial Cells Blood Substitutes and Biotechnology
编者推荐
人类终极挑战:赋予人造细胞生命,全人工细胞实现临近
二十多年来,制造有生命特征的“人造细胞”是科学家执着的追求,而现在有生物学家预测:第一个全人工合成的细胞可能会在十年内出现。如果这个细胞还能成长、分裂,无疑那将是人类巨大的进步。
Cell重磅:十年磨一剑,科学家成功合成出可正常生长分裂的 “人造细胞”
人工细胞的合成对于科学家理解生命与基因的奥秘具有重要意义。20 多年以来,人们一直在尝试在实验室中,通过将一些生物分子拼凑起来从而创造出可以自主控制、代谢、维持生命的人造细胞。而现在,这一努力终于取得了重要突破。近日,来自美国 J. Craig Venter 研究所(JCVI),美国国家标准与技术研究院(NIST)以及麻省理工学院(MIT)的研究人员合作,成功创造出了一个非常简单的人工合成细胞,该细胞可以正常生长和分裂。
本中文词条由小竹凉翻译,薄荷编辑,如有问题,欢迎在讨论页面留言。
本词条内容源自wikipedia及公开资料,遵守 CC3.0协议。